Treatment-Resistant Depression · Patient Decision Guide
Is Spravato or TMS better for treatment-resistant depression?
You’ve tried two antidepressants. Maybe three. Your psychiatrist mentions Spravato. Or TMS. You go home, type the question into Google at 11 PM, and end up on a Reddit thread that’s mostly people arguing.
This is a decision guide for that moment.

Why the question feels impossible to answer
Because the two treatments aren’t actually comparable in the way Google wants them to be. Spravato (esketamine) is a glutamatergic medication you take in a clinic. TMS is a magnetic field that briefly fires a circuit in the front of your brain. Same goal — get your depression to lift when antidepressants haven’t — but they get there through completely different doors.
So the right question isn’t “which is better.” The right question is “which is better for me, given my history, my schedule, my body, and my insurance.” Most of this guide is about answering that.
What each one actually is
Spravato (esketamine)
What it is: a nasal spray version of S-ketamine. FDA-approved for treatment-resistant depression in 2019, and on January 21, 2025, FDA-approved as the first and only monotherapy for TRD — meaning it can now be prescribed without an oral antidepressant alongside it.[1,17,18]
How it works: targets the glutamate system (NMDA receptors), which is a different neurotransmitter pathway than the serotonin and norepinephrine systems your antidepressants have been trying to nudge.[2]
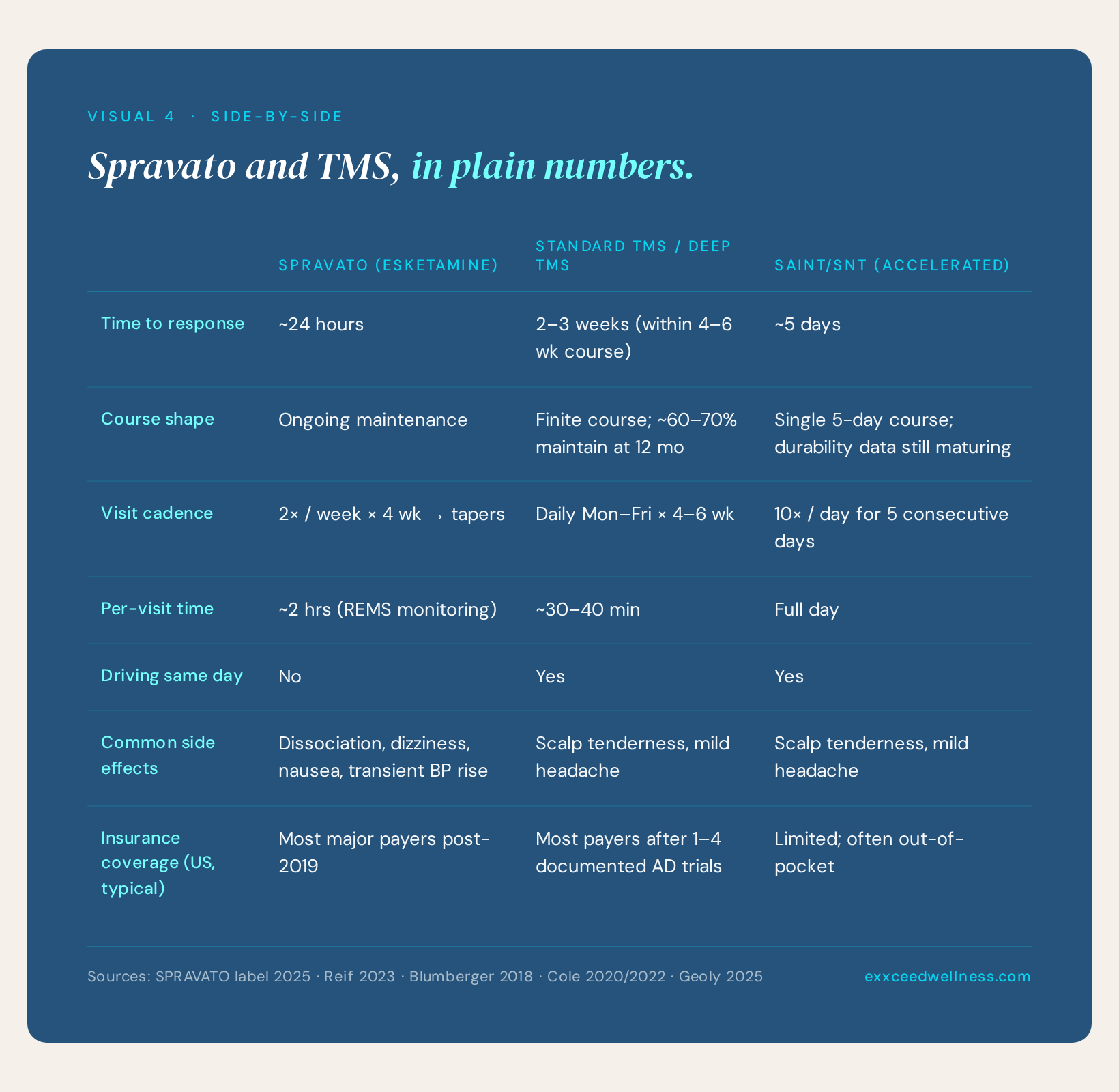
What a session looks like: you arrive at a REMS-certified clinic, take the spray under direct nursing observation, and stay for at least two hours of monitoring while the medication works through your system. The schedule starts at twice weekly for four weeks, then drops to weekly, then to every one-to-two weeks for maintenance. You don’t drive home that day.
TMS (transcranial magnetic stimulation)
What it is: an external device that delivers focused magnetic pulses through your scalp into the dorsolateral prefrontal cortex — the part of the brain that’s underactive in depression. FDA-cleared for depression since 2008, with newer versions adding deeper coils, theta-burst protocols, and accelerated five-day schedules.
How it works: the magnetic pulses depolarize cortical neurons under the coil. Over a course of treatment, the repeated firing recalibrates activity in the brain networks that depression has dialed down.
What a session looks like: you sit in a treatment chair while a tech places the coil against your head and the pulses fire for anywhere from three to forty minutes depending on the protocol. Most patients return to work the same day. The standard course is daily Monday–Friday for four to six weeks. Accelerated protocols (the Stanford SAINT/SNT version) compress that into five consecutive days, with ten sessions per day spaced across the day.
“TMS” in casual conversation usually refers to one of four things: standard 10 Hz repetitive TMS, intermittent theta-burst (iTBS, the three-minute protocol), Deep TMS (the H1 coil that reaches deeper into the brain), or accelerated SAINT/SNT. They share a mechanism but differ in session count, intensity, and the depth of evidence behind them. When your psychiatrist says “TMS,” ask which one.

What the numbers actually say
Here’s where the question gets honest. There is no large head-to-head trial of Spravato versus TMS in treatment-resistant depression. There is one published direct comparison — a small retrospective Italian study of forty patients — and it found accelerated TMS responded faster at one month, but the difference between the two had washed out by six months.[3]
So we triangulate from each treatment’s own data.
Spravato (combined with an oral antidepressant, the original approval pathway):
The largest active-comparator study, ESCAPE-TRD, pitted Spravato against extended-release quetiapine. By week 8, 27.1% of the Spravato arm reached remission versus 17.6% of the quetiapine arm.[4] The pivotal phase 3 trial, TRANSFORM-2, showed a meaningful but modest 4-point improvement on the standard depression rating scale versus antidepressant plus placebo.[5] An independent re-analysis published in 2025 pooled seven Spravato trials and found the effect to be statistically significant but smaller than the manufacturer’s own pre-specified clinical-significance threshold.[6] The honest read: it works, it’s not a magic switch, and your prescriber should set realistic expectations going in.
For long-term maintenance: SUSTAIN-3, the largest extension study with over 3,700 patient-years of exposure, showed about half of patients in remission at the maintenance endpoint and an acceptable safety profile over multiple years.[7]
TMS:
The benchmark non-inferiority trial, THREE-D, randomized 414 TRD patients to either standard 10 Hz rTMS or iTBS. Depression scores fell by roughly half in both groups.[8] The Brainsway Deep TMS pivotal trial showed 38% response and 33% remission with active treatment versus 21% and 15% with sham.[9] Real-world data from the NeuroStar Outcomes Registry — over 5,000 patients across 103 sites — landed at 58–83% response and 28–62% remission, depending on the rating instrument.[10] Of patients who responded acutely, 62.5% were still maintaining that response at one year, and a meaningful share of relapsers responded to a second course.[11]
The Stanford accelerated protocol (SAINT/SNT) generated the most-cited headline number in the field: 86% remission in the original open-label study.[12] The follow-up sham-controlled trial showed a substantially larger response in the active arm than in sham.[13] Twelve-week durability data published in 2025 showed about a third of patients still in remission at three months — meaningful, but a more sober number than the open-label headline implied.[14]
Both treatments help a meaningful share of people who haven’t responded to two or more antidepressants. The remission rates aren’t dramatically different. The differences that matter for you are usually elsewhere.
How fast it works, and how long it lasts
Spravato works fast. Within 24 hours of the first dose, patients in the monotherapy approval trial showed a statistically significant improvement on the depression rating scale compared with placebo.[1] That’s faster than any oral antidepressant can deliver.
Standard TMS doesn’t move that fast. Most patients notice a shift somewhere in weeks two to three of a four-to-six week course. The exception is SAINT/SNT, which is designed to compress the entire course into five days, with the goal of getting patients into remission inside a week. The trade-off there is that durability data are still maturing and access is uneven.
For sustained benefit, the two treatments behave fundamentally differently:
Spravato is ongoing care. Most patients who respond stay on it long-term, with twice-monthly or monthly maintenance dosing. There is no “finish line.” The randomized-withdrawal study, SUSTAIN-1, showed that patients who stayed on Spravato had roughly half the relapse rate of those switched to placebo over the maintenance period.[15]
TMS is a discrete course. You finish it. About 60–70% of acute responders maintain that benefit for a year or more without retreatment, and many of those who relapse respond to a second course.[11]
This is one of the most important distinctions in the whole comparison. If you want a treatment with an ending, TMS is the option that gives you one. If you’re comfortable with ongoing maintenance and want the fastest acute response, Spravato is the option that delivers it.
What it actually feels like
Spravato
The first ninety minutes after dosing is a noticeably altered state. Most patients describe dissociation: a sense of floating, watching themselves from outside, time stretching or feeling unfamiliar. About a quarter to a third of patients report some version of this each session, and it generally fades by the end of the two-hour observation window.[16] Common physical side effects include dizziness, nausea, mild sedation, and a transient blood pressure rise around the forty-minute mark.[16,19]
You don’t drive that day. You’ll need a ride. Plan that into your week before you start.
TMS
You’ll hear a tapping or clicking from the coil. You’ll feel it on your scalp — sometimes mildly uncomfortable in the first few sessions, often barely noticeable by the end of the first week. The most common side effects are scalp tenderness and a mild headache, both of which tend to fade quickly.[8,9]
Seizures are an extremely rare but real risk; the per-course rate is well under 1 in 1,000 with current protocols and proper screening. There’s no sedation, no dissociation, no driving restriction, and no systemic interaction with your other medications.
Cost and access — the realities
Idealism meets the front desk here. A few practical points your prescriber may not get to in a fifteen-minute med-management visit:
Spravato is covered by most major U.S. insurers as of the post-2019 rollout, distributed through specialty pharmacy, and must be administered in a REMS-certified clinic. The practical commitment is roughly eight visits in the first month — twice weekly, two-hour appointments — tapering after week four. The two-hour monitoring is non-negotiable; it’s a federal safety requirement, not a clinic preference. You’ll want to plan child care, ride coverage, and time off work accordingly.
Standard TMS is covered by most insurers after one to four documented antidepressant trials, depending on your plan. The schedule is daily Monday–Friday for four to six weeks (twenty to thirty visits total). No monitoring, no driving restriction, no medication burden during the active course. The friction point is time: patients with rigid schedules or long commutes often can’t make it work.
SAINT/SNT, the five-day accelerated protocol, compresses TMS into a single workweek. Coverage is the rate-limiting issue — it’s newer, and many insurers haven’t fully integrated it yet. Many patients pay out of pocket. If you can take a week off and the access is available, this is the most compressed option of the three.
How to think about which is right for you
There’s no algorithm here, but there is a usable framework. Two questions matter most.

Question 1: How much medication burden do you want to take on?
If you’ve already cycled through multiple medications and you’re tired of the trial-and-error, TMS may appeal. It’s medication-free during the active course, finite, and doesn’t add to your prescription list. You walk in, finish, walk out.
If you’re comfortable with the medication frame and want something that works fast — within days — Spravato is the option that delivers the fastest acute response.
Question 2: How much time can you give to the front-loaded burden?
Standard TMS asks for four to six weeks of weekday visits. Patients with flexible work or who live close to a clinic find it manageable; patients whose schedules can’t bend often can’t make the math work.
Spravato is initially eight two-hour visits in the first month, then it tapers — but it doesn’t end the way a TMS course ends. Different burden, different shape, and a different end-state.
SAINT/SNT, if access and coverage allow, compresses the TMS commitment into a single week. It’s the highest-burden week of the three options, and the lowest-burden month.

So which is “better”?
The honest answer: both are real options for treatment-resistant depression, with imperfect but meaningful evidence behind them. The retrospective head-to-head data we have suggest they may converge by six months.[3] The remission rates from each treatment’s own trials are in roughly the same neighborhood, even if the trial designs make a clean comparison impossible.
What separates them isn’t usually efficacy. It’s how fast you need relief, how much time you can give to weekday visits, whether you want a finite course or ongoing maintenance, whether you want medication-free treatment, what your insurance will cover, and small practical things like having a ride home.
Patients tend to do well with this decision when they stop framing it as a winner-take-all leaderboard and start framing it as which fits their life and their wiring. The “right” answer is the one you can actually start, complete, and live around.
How to start
If you’re considering either, the next step is a treatment-resistant depression evaluation: a clinician with experience in both interventions reviewing your full medication history (which doses, for how long, what worked partially, what didn’t), confirming you meet TRD criteria, and walking you through the practical realities of each option. That’s where a yes/no for either treatment becomes a plan — sequenced, paid for, and tied to your real life.
Two failed antidepressants is not the end of the road. It’s the threshold for a different conversation.
References
- Janik AM, Doherty T, Bandinelli PL, et al. Esketamine monotherapy in adults with treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2025;82(9):877–887. doi:10.1001/jamapsychiatry.2025.1317 [PMID 40601310]
- McIntyre RS, Lui LMW, Mansur RB, et al. Glutamatergic modulators for major depressive disorder. CNS Drugs. 2024;38(11):869–890. doi:10.1007/s40263-024-01114-y [PMID 39150594]
- Prato A, Comai S, Bellani M, et al. Real-world comparison of esketamine nasal spray and accelerated repetitive transcranial magnetic stimulation in treatment-resistant depression. Int J Psychiatry Clin Pract. 2026. doi:10.1080/13651501.2026.2642680 [PMID 41814584]
- Reif A, Bitter I, Buyze J, et al. Esketamine nasal spray versus quetiapine for treatment-resistant depression. N Engl J Med. 2023;389(14):1298–1309. doi:10.1056/NEJMoa2304145 [PMID 37792613]
- Popova V, Daly EJ, Trivedi M, et al. Efficacy and safety of flexibly dosed esketamine nasal spray combined with a newly initiated oral antidepressant in treatment-resistant depression: a randomized double-blind active-controlled study. Am J Psychiatry. 2019;176(6):428–438. doi:10.1176/appi.ajp.2019.19020172 [PMID 31109201]
- Naudet F, Maria AS, Falissard B. Esketamine for treatment-resistant depression: an individual-patient-data meta-analysis. BMC Med. 2025;23(1):677. doi:10.1186/s12916-025-04435-x [PMID 41310599]
- Zaki N, Chen LN, Lane R, et al. Long-term safety and efficacy of esketamine nasal spray in treatment-resistant depression: SUSTAIN-3 final analysis. Int J Neuropsychopharmacol. 2025;28(6). doi:10.1093/ijnp/pyaf027 [PMID 40319349]
- Blumberger DM, Vila-Rodriguez F, Thorpe KE, et al. Effectiveness of theta burst versus high-frequency repetitive transcranial magnetic stimulation in patients with depression (THREE-D): a randomised non-inferiority trial. Lancet. 2018;391(10131):1683–1692. doi:10.1016/S0140-6736(18)30295-2 [PMID 29726344]
- Levkovitz Y, Isserles M, Padberg F, et al. Efficacy and safety of deep transcranial magnetic stimulation for major depression: a prospective multicenter randomized controlled trial. World Psychiatry. 2015;14(1):64–73. doi:10.1002/wps.20199 [PMID 25655160]
- Sackeim HA, Aaronson ST, Carpenter LL, et al. Clinical outcomes in a large registry of patients with major depressive disorder treated with transcranial magnetic stimulation. J Affect Disord. 2020;277:65–74. doi:10.1016/j.jad.2020.08.005 [PMID 32799106]
- Dunner DL, Aaronson ST, Sackeim HA, et al. A multisite, naturalistic, observational study of TMS for patients with pharmacoresistant major depressive disorder: durability of benefit over a 1-year follow-up period. J Clin Psychiatry. 2014;75(12):1394–1401. doi:10.4088/JCP.13m08977 [PMID 25271871]
- Cole EJ, Stimpson KH, Bentzley BS, et al. Stanford Accelerated Intelligent Neuromodulation Therapy for treatment-resistant depression. Am J Psychiatry. 2020;177(8):716–726. doi:10.1176/appi.ajp.2019.19070720 [PMID 32252538]
- Cole EJ, Phillips AL, Bentzley BS, et al. Stanford Neuromodulation Therapy (SNT): a double-blind randomized controlled trial. Am J Psychiatry. 2022;179(2):132–141. doi:10.1176/appi.ajp.2021.20101429 [PMID 34711062]
- Geoly AD, Bishop JH, Gulser M, et al. Stanford Neuromodulation Therapy: durability of remission in treatment-resistant depression. Brain Stimul. 2025;18(3):875–881. doi:10.1016/j.brs.2025.04.006 [PMID 40209894]
- Daly EJ, Trivedi MH, Janik A, et al. Efficacy of esketamine nasal spray plus oral antidepressant treatment for relapse prevention in patients with treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2019;76(9):893–903. doi:10.1001/jamapsychiatry.2019.1189 [PMID 31166571]
- Wajs E, Aluisio L, Holder R, et al. Esketamine nasal spray plus oral antidepressant in patients with treatment-resistant depression: assessment of long-term safety in a phase 3, open-label study (SUSTAIN-2). J Clin Psychiatry. 2020;81(3):19m12891. doi:10.4088/JCP.19m12891 [PMID 32316080]
- U.S. Food and Drug Administration. Approval letter, NDA 211243 supplement (S-018), SPRAVATO (esketamine) monotherapy for treatment-resistant depression. January 21, 2025. accessdata.fda.gov
- Johnson & Johnson. SPRAVATO (esketamine) approved in the U.S. as the first and only monotherapy for adults with treatment-resistant depression [press release]. January 21, 2025. jnj.com
- SPRAVATO (esketamine) U.S. prescribing information. Revised 2025. accessdata.fda.gov label
This article is for educational purposes and does not replace evaluation by a licensed clinician. Patients with treatment-resistant depression should discuss their specific history and options with a qualified psychiatric provider before initiating any new treatment.


Comments are closed